Almost a year after the blockbuster weight loss drug Mounjaro, manufactured by pharma giant Eli Lilly, was launched in India, a major counterfeit syndicate allegedly dealing in fake versions of the drug has been busted in Gurgaon.
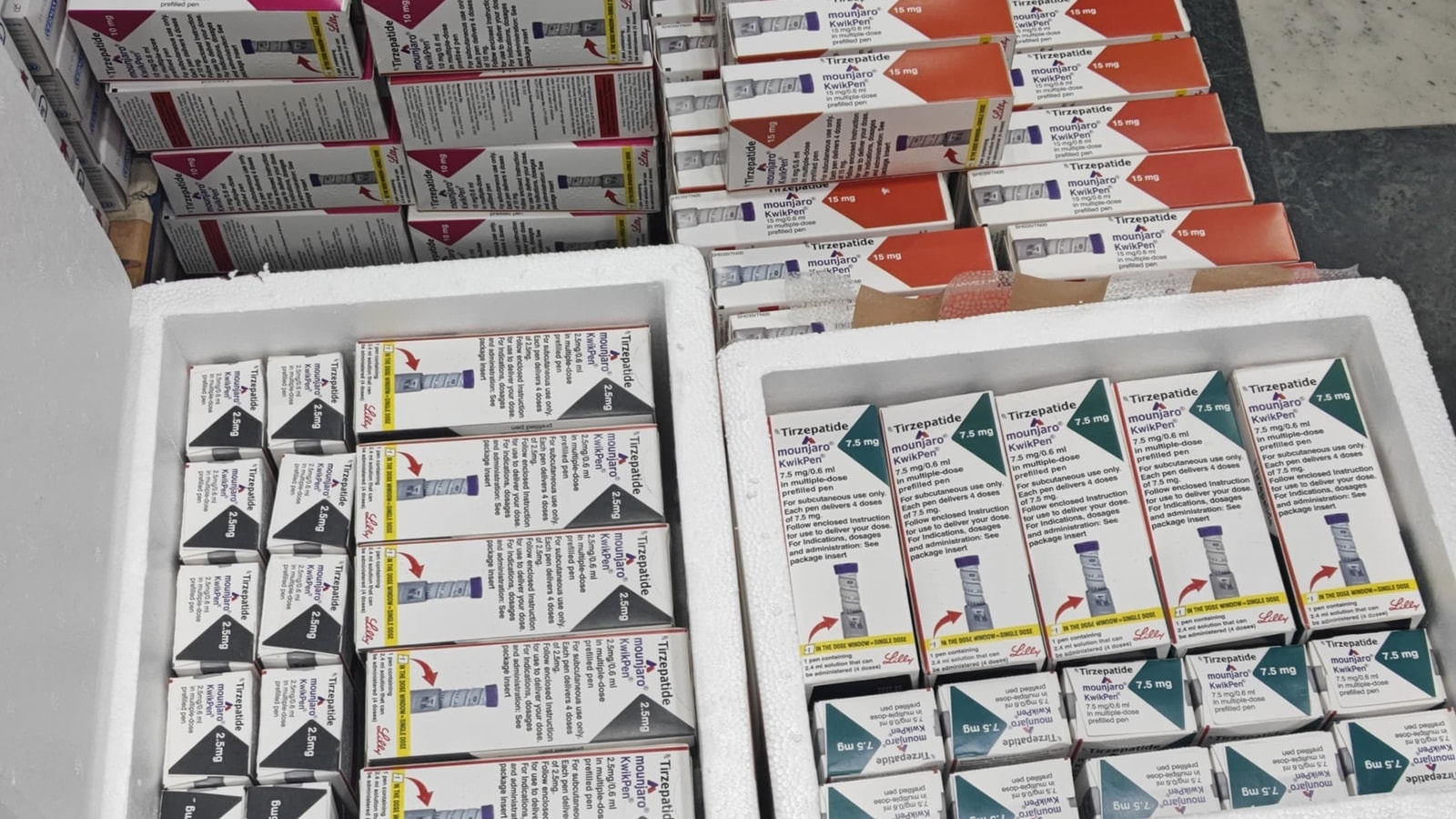
On Sunday, police in Gurgaon said spurious Mounjaro injections — used for diabetes management and weight loss — valued at over Rs 56 lakh were recovered from a car. The case marks one of the first alleged counterfeit rackets reported in India involving GLP-1 weight-loss drugs, a new class of medications that entered the Indian market last March.
Around 7:25 pm on Saturday, a team of the Drugs Control Office in Gurgaon intercepted a Swift Dzire, operating as a cab, in DLF Phase-IV near Millennium City Centre Station after receiving a tip-off about the alleged sale of spurious medicines, said officers. The man inside the vehicle identified himself as Mujammil Khan and said he was a medical representative.
During questioning, Khan alleged that the entire illicit stock recovered from the cab belonged to one Avi Sharma (32), according to a spot memo prepared by Drugs Control Officer Amandeep Chauhan. As both Sharma and Khan were gradually arrested, it was revealed during the probe that Sharma allegedly produced the entire spurious stock at his home in Gurgaon. The memo noted, “…Sharma was asked to provide the details of where he sold the products, to which he did not cooperate at the spot.”
“As of now Sharma seems to be only one who ran this entire operation,” said Chauhan, adding, “As per the initial questioning, he started the illegal operation on April 2. We are questioning him further to know the supply chain and to whom and how he was selling. From his residential premises, we recovered another Rs 14 lakh worth of such spurious injections.”
Sharma, a BBA graduate from a university in Gurgaon, worked at his father’s firm till 2016, said officers, adding that he claimed to have then founded an Ayurvedic company, Hemped Souls International LLP, after that. Khan, when he was stopped by the team, told the police that he was a medical representative at Hemped.
Mounjaro’s demand, health officials note, has surged sharply in the recent months due to off-label use for weight loss, creating a lucrative parallel illegal market.
Story continues below this ad
As per the memo, after the cab with Khan was intercepted on Saturday, Sharma was called to the location and he arrived around 12:10 am on Sunday. When asked to produce a valid licence of production, Sharma “informed that he is having no licence in this name and he is solely responsible for his Act,” the official record noted.
The document further stated that Sharma allegedly disclosed “that he himself manufactured the product in question from his premises” located at Pivotal Paradise at Sector 62 of Gurgaon. Khan and Sharma were arrested on the spot for violating the Drugs and Cosmetics Act, as per the document.
The estimated cost of the sampled injection strengths (ranging from 2.5 mg to 15 mg) was calculated to be ₹56,15,847 based on MRP, according to the memo.
Form 17 attached with the memo itemised the seizure of a total of 242 Mounjaro KwikPen injections across six different dosage strengths.
Story continues below this ad
Following the seizure, an official order by the Drugs Control Officer recommended a halt on the commercial distribution of the flagged batches.
The order stated that “in the opinion of undersigned if agreed by the State Drugs Controller, Haryana, the present matter be forwarded to competent authority so that the sale of above batches of the product in question be immediately stopped till the verification is not conducted…”
The spot memo detailed that physical verification of the recovered stock against digital labels provided by the official importer revealed multiple discrepancies, and had typographical errors.
A spokesperson for Eli Lilly and Company (India) Monday evening issued the following statement:
Story continues below this ad
“We have been made aware of a recent development in relation to the seizure of a suspicious and counterfeit products that allegedly carry our product brand name Mounjaro (Tirzepatide). The said seizure resulted from an enforcement drive conducted in Gurugram by officials from the state drugs regulatory authority, Haryana.
Lilly takes patient safety extremely seriously and welcomes regulatory authority’s action against illicit medicines. We are actively supporting the investigation and will continue to work with regulatory and law enforcement authorities worldwide to protect patients from the risks of counterfeit products. Stronger, coordinated enforcement must be sustained if we are to protect patients from unsafe fake medicines.”